Jointechlabs is an emerging leader in point-of-care regenerative medicine therapies worldwide.
FDA-cleared: MiniTC® for microfat.
CE-Mark approved: Mini-Stem System® for SVF processing outside the USA.

Brilliant technology for Microfat
MiniTC® Adipose Processing System
MiniTC® is a device that processes autologous fat (adipose tissue) into purified, finely washed Microfat for injection.

What is microfat and why is it used?
Greater benefits, fewer complications
Microfat is an autologous biologic with important advantages over synthetic products and other biologics.
Learn More
Many uses for active, pain-free life
Microfat therapies using MiniTC®
Pain management, orthopedics, sports medicine, aesthetics, reconstruction, and wound healing.
Learn More
Benefits patients, doctors, & practices
Win–win–win for all participants
Better healing without side effects. Satisfied patients and clinic staff. Greater return for doctors.
Learn More
Tell us your thoughts, ask a question, or start a discussion.

MiniTC® Advantages
MiniTC® is unique in its combination of high-quality output, convenience, simplicity, price, and adherence to regulatory standards.
- In-Clinic Use
- All-in-one Box
- FDA-Cleared
- Simplicity and Speed
- Highest Cell Count and Viability


Free infographic on fat and microfat!
Understand the miracle of fat.
View on screen, or print and display in a waiting or exam room.
For Patients
We love your fat
Ask your doctor about how MiniTC® and microfat can help with:

Want to Learn More About the Benefits of the MiniTC® Adipose Processing System?

Testimonials
Events

Jointechlabs – Silver Sponsor at the Annual Regenerative Medicine Essentials (RME) Course
Date: June 2, 2026
Location: Winston-Salem, NC
Read more
ESSKA Congress 2026 | Jointechlabs in Prague Where orthopaedics meets innovation | 20–22 May
Date: May 21, 2026
Location: Prague
Read more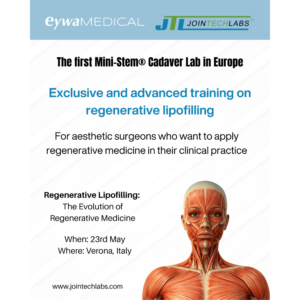
Exclusive and advanced training on regenerative lipofilling
Date: May 23, 2026
Location: ICLO - Verona, Italy
Read more

